Therefore, there are various non-equivalent definitions of atomic radius. Elements with multiple ionic charges are also mentioned in this table. Ionic Charges of all Elements (List) List of elements with their common ionic charges are mentioned below. However, this assumes the atom to exhibit a spherical shape, which is only obeyed for atoms in vacuum or free space. When atoms gain electron/s, the negatively charged ion is formed, and when the atoms lose electron/s, the positively charged ion is formed. The atomic radius of a chemical element is a measure of the distance out to which the electron cloud extends from the nucleus. It must be noted, atoms lack a well-defined outer boundary. The atomic radius of Carbon atom is 69pm (covalent radius). Note that, each element may contain more isotopes, therefore this resulting atomic mass is calculated from naturally-occuring isotopes and their abundance. 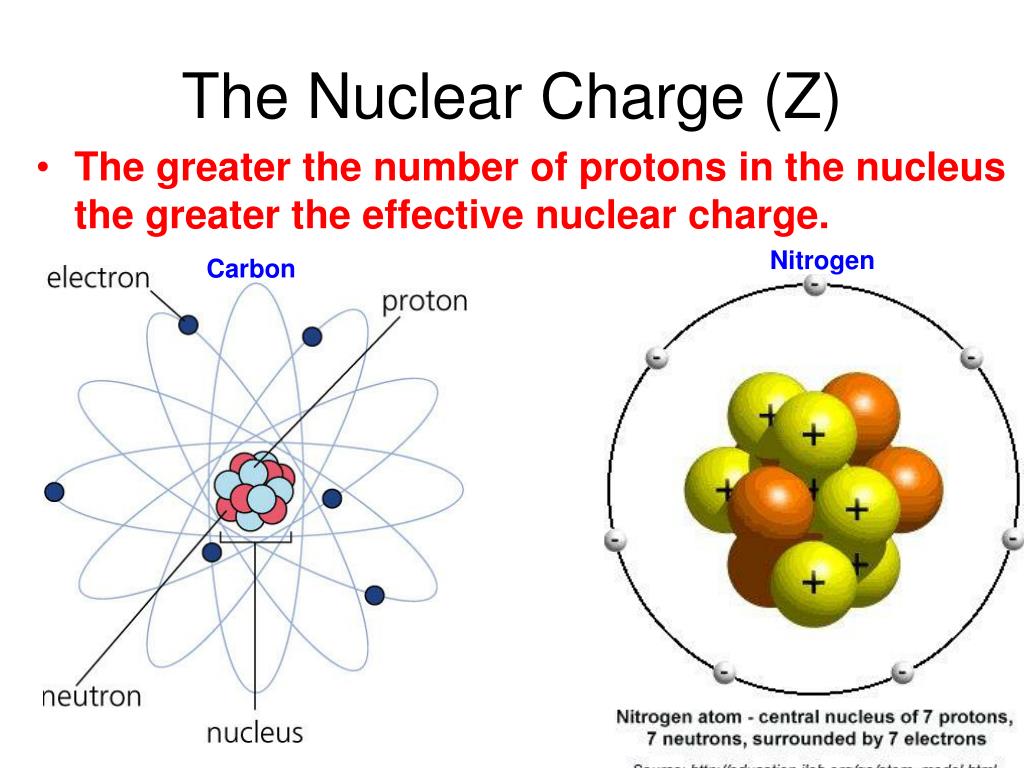
The atomic mass is carried by the atomic nucleus, which occupies only about 10 -12 of the total volume of the atom or less, but it contains all the positive charge and at least 99.95% of the total mass of the atom. The atomic mass or relative isotopic mass refers to the mass of a single particle, and therefore is tied to a certain specific isotope of an element. Mass numbers of typical isotopes of Carbon are 12 13. Isotopes are nuclides that have the same atomic number and are therefore the same element, but differ in the number of neutrons. Animation of a Carbon atom that shows charges of electrons, protons, and neutrons, When building an atom. The difference between the neutron number and the atomic number is known as the neutron excess: D = N – Z = A – 2Z.įor stable elements, there is usually a variety of stable isotopes. The Atom Builder Guide to Building a Stable Atom. Neutron number plus atomic number equals atomic mass number: N+Z=A. The total number of neutrons in the nucleus of an atom is called the neutron number of the atom and is given the symbol N. The total electrical charge of the nucleus is therefore +Ze, where e (elementary charge) equals to 1,602 x 10 -19 coulombs. Total number of protons in the nucleus is called the atomic number of the atom and is given the symbol Z. NOAA atmospheric scientists use these isotopic labels to determine what percent of that carbon was derived from fossil fuels, the terrestrial biosphere, or from the ocean.Atomic Number – Protons, Electrons and Neutrons in CarbonĬarbon is a chemical element with atomic number 6 which means there are 6 protons in its nucleus. A defect-rich porous carbon (K-defect-C-1100) was synthesized by a K +-assisted strategy (Ling et al., 2022) and has 12-vacancy-type defects (V 12) (Figures 5AD). These differences in isotopic abundance are used as “labels” to identify the different sources of CO 2 found in an atmospheric CO 2 sample. In addition to heteroatom doping, the construction of intrinsic carbon defects causes charge delocalization, activating carbon atoms at defect sites for better catalytic properties.  
Certain isotopes are more abundant in some materials than others since some physical and chemical processes “prefer” one isotope over another. Although isotopes of the same element are twins when it comes to reactivity, the different number of neutrons means that they have a different mass. Isotopes are atoms of the same element that have a different number of neutrons. Neutrons are neutral - they have no charge. Carbon Energy is an Open Access journal: authors of accepted articles pay an article publication charge, and their articles are published under a Creative. For example, a carbon atom has six protons, but an atom with only five protons is boron while an atom with seven protons is the element nitrogen. The Federal Fuel Charge (sometimes referred to as the Federal Carbon Tax by retailers on utility bills) was implemented in Alberta as of January 1, 2020. In other words, the number of protons is what gives each element its unique, individual identity. Protons are positively charged particles, and the number of protons is always fixed for a particular element. The small, dense nucleus (or center) of the atom contains the other components–the protons and neutrons. However, the atom remains the same element whether it has a positive, negative, or neutral charge. Atoms may gain or lose electrons, which change the charge of the atom (creating ions). Although atoms are too small to see without using high-powered microscopes, they are composed of even smaller particles: protons, neutrons, and electrons.Įlectrons, which are extremely light, negatively-charged particles, orbit around a central mass–the nucleus of an atom. The Technical Details: Chemistry Composition of an AtomĪtoms, which are the basic, fundamental unit of all matter, can differ greatly from one another. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
